| . |  |
. |
| . |  |
. |
|
by Staff Writers New Haven CT (SPX) Oct 23, 2019
The search for sustainable approaches to generating new fuels has brought scientists back to one of the most abundant materials on Earth - reddish iron oxide in the form of hematite, also known as rust. Researchers say rust has long been seen as a potentially attractive material for solar water splitting, a key process that plants employ in photosynthesis. Plants use an enzyme called photosystem II (PSII) to absorb light and split water, extracting electrons and protons from the water molecules and generating the atmospheric oxygen that sustains life on Earth. Rust may offer a way to mimic the solar water splitting aspect of photosynthesis, enabling fuel to be generated from water, either by combining protons and electrons for hydrogen generation, or by using the electrons and protons to convert carbon dioxide into hydrocarbon fuels. The problem, scientists say, is that rust's performance in numerous water-splitting experiments has been disappointing compared to its theoretical potential. They say a fundamental understanding of the reaction mechanism is lacking, preventing the development of devices for direct solar water splitting. A new study by Victor Batista at Yale University in collaboration with James Durrant from Imperial College, London, and Michael Gratzel from the Institut des Sciences et Ingenierie Chimiques, Ecole Polytechnique Federale de Lausanne, Lausanne, Switzerland, offers some answers. The study sheds light on the mechanism of water oxidation occurring at the metal oxide/water interface. A combined computational and experimental study on the water oxidation mechanism found that if you change the intensity of light, you also change the mechanism of water splitting on hematite. The researchers say this provides valuable hints on the nature of the sites responsible for reactivity on the oxide surface. Computational work performed by study co-author Ke Yang, a postdoctoral researcher in the Batista group, identified isolated catalytic sites - under low-light intensity and a two-atom, Fe(OH)-O-Fe(OH) catalytic core - that build enough oxidation power to extract electrons from water by accumulating up to three oxidizing equivalents (missing electrons) under 1 sun operating conditions (the full intensity of sunlight on a clear, bright day). This mechanism mimics the activation of PSII during photosynthesis, the researchers said. "The integration of computational and experimental work has been essential for elucidating the nature of the catalytic sites on rather complicated metal oxide surfaces and the dependency of the reaction mechanism under low- and high-light intensity conditions," said Batista. In 2018, Batista co-authored a separate study that described similar, two-atom catalysts. The researchers said that finding, together with the new study, suggests that two-atom catalytic cores featuring two adjacent metal centers might be particularly suited to achieve efficient water splitting. "Making oxygen from water requires multiple oxidations," Durrant said. "Experimentally, the key to our study has been using optical absorption spectroscopy to measure how the kinetics of water oxidation change as we accumulate more holes on the hematite surface. This has allowed us to determine rate laws and rate constants for the reaction; for example, determining how many holes have to come together to access the rate limiting step of the reaction, and determining the activation energy for the reaction."
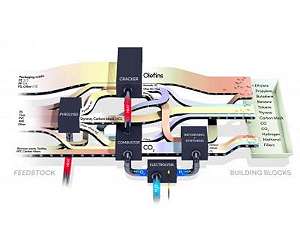
Turning plastic waste back into high-quality plastic with advanced steam cracking Gothenburg, Sweden (SPX) Oct 19, 2019 A research group at Chalmers University of Technology, Sweden, has developed an efficient process for breaking down any plastic waste to a molecular level. The resulting gases can then be transformed back into new plastics - of the same quality as the original. The new process could transform today's plastic factories into recycling refineries, within the framework of their existing infrastructure. The fact that plastics do not break down, and therefore accumulate in our ecosystems, is one of our ... read more
|
|||||||||||||
| The content herein, unless otherwise known to be public domain, are Copyright 1995-2026 - SpaceDaily. All websites are published in Australia and are solely subject to Australian law and governed by Fair Use principals for news reporting and research purposes. By using our websites you consent to cookie based advertising. If you do not agree with this then you must stop using the websites from May 25, 2018. Privacy Statement. Additional information can be found here at About Us. |